Sulfurous acid, calcium salt
Calcium sulfite
CAS: 10257-55-3
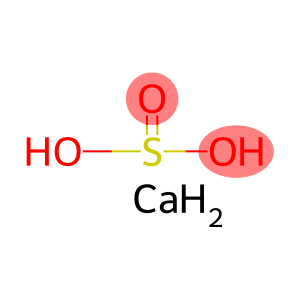
Molecular Formula: CaO4S
Sulfurous acid, calcium salt - Names and Identifiers
| Name | Calcium sulfite |
| Synonyms | Calciumsulfit Calcium sulfite calcium sulphite CalciumSulfitePowder Sulfurous acid, calcium salt |
| CAS | 10257-55-3 |
| EINECS | 233-596-8 |
| InChI | InChI=1/Ca.H2O3S/c;1-4(2)3/h;(H2,1,2,3)/q+2;/p-2 |
Sulfurous acid, calcium salt - Physico-chemical Properties
| Molecular Formula | CaO4S |
| Molar Mass | 136.1406 |
| Density | 33.3[at 20℃] |
| Melting Point | 600°C |
| Water Solubility | 0.0043g/100mL (18°C), 0.001g/100mL (100°C) H2O; slightly soluble alcohol; soluble in acid solutions with SO2 evolution [MER06] [CRC10] |
| Appearance | White solid |
| Storage Condition | Room Temprature |
| Use | Used as analytical reagents, also used in textile, paper and sugar industry |
Sulfurous acid, calcium salt - Introduction
Lose crystal water at 100 ℃. Decompose at 650 ℃. Slightly soluble in water. Slowly oxidized to calcium sulfate in the air. Decompose in acid to release sulfur dioxide.
Last Update:2022-10-16 17:11:35
Sulfurous acid, calcium salt - Reference Information
| EPA chemical information | Information provided by: ofmpub.epa.gov (external link) |
| application | calcium sulfite can remove residual chlorine in water, including chemical residual chlorine NH2CL,NHCL2 and NCL3 and free residual chlorine CLO-,HOCL,CL2, etc. Compared with traditional activated carbon, the chlorine removal effect is more efficient, safe, high temperature resistant, and no bacteria breeding, it is widely used in various water to remove residual chlorine in Southeast Asian countries and regions such as Japan, South Korea, Malaysia, and Taiwan. |
| Use | Used as analytical reagent, also used in textile, papermaking and sugar industries, etc. Used as analytical reagent, also used in making calcium and plastic materials, and also used as cellulose products Bleaching dechlorination agent, food preservative, fermentation fungicide, etc. |
Last Update:2024-04-09 19:05:09
Supplier List
Featured ProductsSpot supply
Product Name: Calcium sulfite Visit Supplier Webpage Request for quotationCAS: 10257-55-3
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Calcium sulfite Request for quotation
CAS: 10257-55-3
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 10257-55-3
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: Calcium sulfite Request for quotation
CAS: 10257-55-3
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
CAS: 10257-55-3
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
Product Name: Calcium sulfite Request for quotation
CAS: 10257-55-3
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
CAS: 10257-55-3
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
Product Name: Calcium sulfite Visit Supplier Webpage Request for quotation
CAS: 10257-55-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 10257-55-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Featured ProductsSpot supply
Product Name: Calcium sulfite Visit Supplier Webpage Request for quotationCAS: 10257-55-3
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Calcium sulfite Request for quotation
CAS: 10257-55-3
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 10257-55-3
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: Calcium sulfite Request for quotation
CAS: 10257-55-3
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
CAS: 10257-55-3
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
Product Name: Calcium sulfite Request for quotation
CAS: 10257-55-3
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
CAS: 10257-55-3
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
Product Name: Calcium sulfite Visit Supplier Webpage Request for quotation
CAS: 10257-55-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 10257-55-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History




